Pharma and biotech organizations invest billions in R&D every year — yet most still struggle to answer a simple question:
Where are our lab assets, and how are they performing?
Across the industry, equipment data lives in spreadsheets, vendor portals, LIMS, ERPs, and email threads. No single system gives IT, Finance, or Lab Operations a reliable view of what’s active, under maintenance, or sitting idle. The result is wasted capital, frustrated scientists, and constant firefighting between departments.
In 2025, solving lab asset tracking isn’t about buying more software — it’s about unifying the stack you already have.
The Real Cost of Broken Lab Asset Tracking
Most R&D organizations don’t have a utilization problem — they have a visibility problem.
When asset records are scattered, three things happen:
- Duplicate purchases and “ghost” inventory: Without a centralized view, teams re-buy instruments they already own, and service contracts remain open for retired units. Ontoforce (2025) notes that “without a transparent system to track instruments and equipment, companies often struggle with duplicate purchases and underutilized assets.”
- Reactive maintenance and downtime: Missed calibrations and late service events stall experiments and create audit risk. McKinsey’s QC-lab study found that digital scheduling and analytics reduce deviations and improve right-first-time performance.
- Finance friction: Every quarter, finance teams ask lab managers to confirm which assets are still active. That “asset validation” cycle can take weeks, draining time from both sides.
The irony is that while most R&D data has gone digital, lab infrastructure data remains analog — trapped in Excel or PDF reports.
Why Traditional Tools Don’t Solve It
Every tool in the lab ecosystem does part of the job — but none handle the full lifecycle:
| System | What it does well | Where it falls short |
| LIMS / ELN | Track experiments, samples, and users | Local schedulers, not enterprise-wide; weak link to finance or IT governance. |
| ERP / Procurement | Manage purchasing and cost centers | No visibility into usage, calibration, or downtime. |
| ServiceNow / ITSM | Track enterprise assets and service workflows | Needs lab semantics — utilization, calibration, GLP workflows. |
| Vendor Portals | Offer rich diagnostics for a single brand | Siloed; can’t unify mixed instrument fleets. |
| Excel / Email | Fast to start | Impossible to audit or scale; no analytics or automation. |
As a result, teams spend their time reconciling data instead of acting on it.
What Modern Asset Tracking Looks Like
Next-generation labs are adopting an integrated enterprise model – combining scientific systems (ELN/LIMS) with operational backbones (EAM/ITSM) and analytics.
According to McKinsey’s Next-Gen Tech Stack for Biopharma R&D (2025), modern R&D organizations are building “AI-ready data pipelines” that connect scientific and operational layers, unlocking predictive maintenance, scheduling optimization, and portfolio-level planning. The key is a system of record for lab assets – one source of truth that tracks every instrument’s status, location, maintenance history, and cost center.
A Practical Roadmap for R&D Leaders
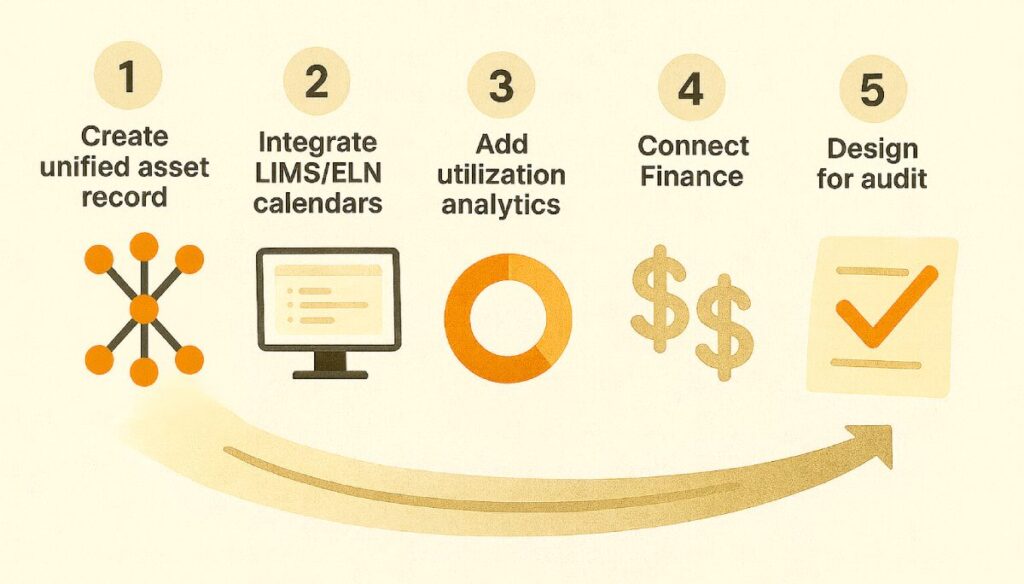
- Create a unified asset record.
Establish an enterprise system as the authoritative source for every lab asset — ownership, warranty, compliance, and location. - Integrate your LIMS/ELN calendars.
Keep scientific context where it belongs, but push bookings, downtime, and “asset unavailable” events into the enterprise workflow layer for maintenance and calibration. - Add utilization analytics.
Aggregate telemetry and scheduling data across all vendors. Use analytics to redeploy idle assets, retire obsolete ones, and plan new purchases based on real use. - Connect Finance.
Link the asset record to ERP cost centers and budgets. Automate chargebacks and allocations based on usage rather than manual reconciliation. - Design for audit from the start.
Centralize calibration and service logs with digital signatures and immutable records. Make “asset readiness” visible at the point of booking.
Proof That It Works
Agilent’s Data-Driven Laboratory case study showed how portfolio-level analytics enabled companies to retire and sell underused instruments – freeing capital for new technology.
Similarly, CrossLab Connect demonstrated that comparing utilization to repair metrics helps identify problematic assets and reduce downtime.
These aren’t abstract benefits. They translate directly into faster science, less waste, and better CapEx discipline.
Where newLab® Fits
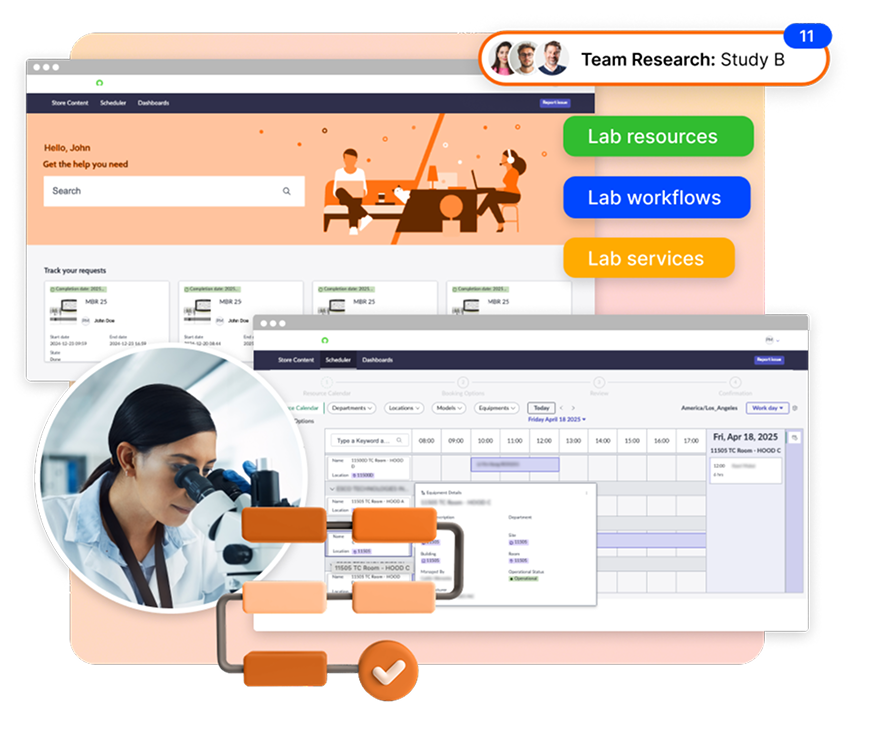
At newLab®, we built our platform natively on ServiceNow so pharma and biotech R&D teams can manage lab operations – scheduling, maintenance, service requests, and assets – all within their existing enterprise ecosystem.
Instead of adding yet another system, newLab®:
- Extends ServiceNow to include lab-specific semantics (calibration, lab workflows, utilization).
- Centralizes asset data from LIMS, ERP, and vendor portals into one layer.
- Provides real-time dashboards for equipment availability, cost, and maintenance.
- Delivers audit-ready records automatically – no more hunting through spreadsheets.
For IT, it means governance and scalability.
For Lab Operations, it means scientists get time back.
For Finance, it means visibility without the manual chase.
The Future of R&D Efficiency
Automation and AI are powerful drivers of innovation – but they can’t deliver their full potential without connected, trustworthy data.
Pharma’s next efficiency breakthrough won’t come from adding more robots or algorithms; it will come from integrating lab data across equipment, scheduling, maintenance, and finance into a single, reliable ecosystem.
When every system speaks the same language, decisions become faster, smarter, and more confident — unlocking true operational intelligence.
As Deloitte’s 2025 research notes, biopharma labs are moving “from siloed to fully integrated, predictive environments.”That’s precisely the transformation newLab® is built to accelerate.
Learn More
See how global R&D organizations are gaining full visibility into their lab assets and eliminating wasted spend.
👉 Book a demo or watch our on-demand webinar: Modernizing Lab Operations in 2025 — How to Eliminate Bottlenecks and Scale R&D with Confidence.
Sources:
- Deloitte — Future-proofing Pharma R&D Labs (2025)
- McKinsey — Next-Gen Tech Stack for Biopharma R&D (2025); Automation in R&D Labs (2023)
- Agilent CrossLab — Data-Driven Laboratory case studies
- Ontoforce (2025) — Inventory Oversight Challenges
- ServiceNow Life Sciences & Community (2024)
- Lab Manager — Leveraging ELNs/LIMS for Asset Scheduling (2023)



