Best Biotechnology Lab Management Software (2026 Guide)
Running a biotechnology lab with 50+ scientists is no small feat. As your research scales, so do the headaches: missing reagents, unbooked or double-booked instruments, inconsistent data, and compliance risks. Studies show scientists spend up to 25% of their time on manual record-keeping and inventory management – time that should go into discovery.
The right lab management software solves this by centralizing data, streamlining workflows, and ensuring regulatory compliance (FDA 21 CFR Part 11, ISO, GMP). Yet, with dozens of options on the market – from ELNs to full LIMS – it’s hard to know where to start.
This guide compares the 7 best biotechnology lab management software solutions for 2026, with an emphasis on:
- Large R&D teams (50+ scientists)
- Compliance-readiness (FDA, ISO, GxP)
- Data integrity & collaboration
- Equipment & resource scheduling
We’ll also show you how newLab complements existing ELN or LIMS tools – filling a critical gap in equipment management and operational efficiency – and how to get a demo.
Comparison Table
| Software | Best For | Core Features | Compliance | Scalability | Pricing Model |
| Benchling | Modern biotech R&D | ELN + LIMS, sequence design, collaboration | 21 CFR Part 11 | High | SaaS, per user |
| Labguru | Small/medium labs | ELN + inventory + equipment tracking | 21 CFR Part 11 | Medium | Subscription tiers |
| LabWare LIMS | Large regulated labs | Full LIMS + ELN, automation | 21 CFR Part 11, GLP, GxP | Very high | License + customization |
| STARLIMS | Pharma/clinical compliance | Enterprise LIMS + analytics | 21 CFR Part 11, GMP | Very high | Enterprise pricing |
| SciNote | Budget-conscious & open source | Open-source ELN + inventory | 21 CFR Part 11-ready | Medium | Free + premium |
| LabVantage | Enterprise informatics | LIMS + ELN + analytics | GxP, FDA | Very high | Subscription + services |
| newLab | Equipment & operations | Scheduling, usage tracking, maintenance, AI | ISO/FDA data trails | High | SaaS (per user) |
The 7 Best Biotechnology Lab Management Software Tools
1. newLab
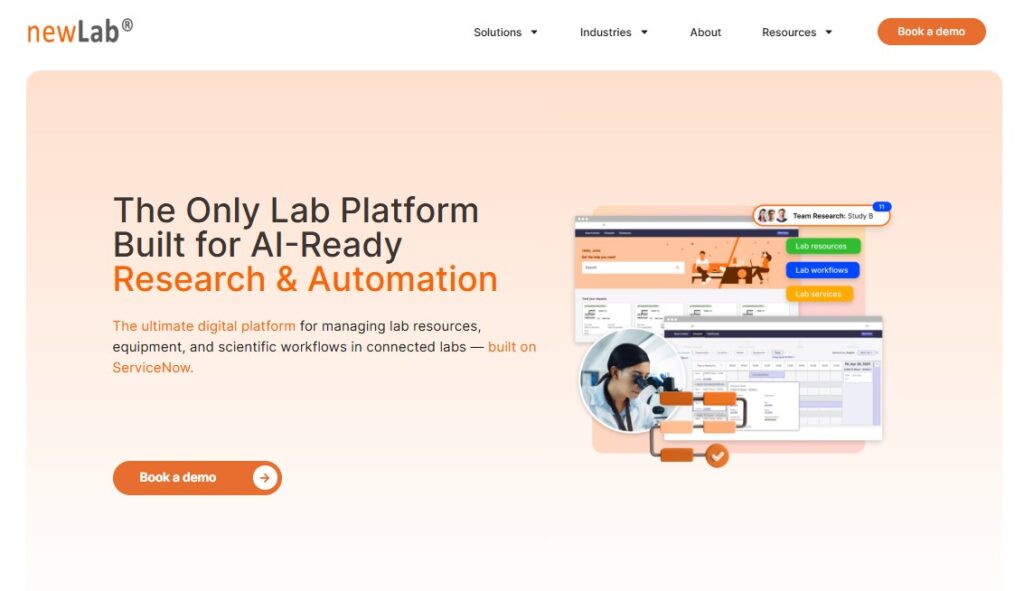
Best for Equipment Management & AI-Ready Operations
Most ELN and LIMS platforms handle data and samples, but struggle with equipment logistics. That’s where newLab shines.
Key features
- Centralized equipment booking & scheduling
- Usage tracking and reporting
- Maintenance & calibration workflows
- Integration with ServiceNow (IT, procurement, incident management)
- AI-ready insights: predict maintenance, optimize resource allocation
Why labs choose newLab
- Solves the shared instrument chaos – no more double-bookings or manual calendars
- Seamless compliance audit trails for equipment
Integrates into existing ELN/LIMS ecosystems - Scales easily with growing R&D teams
Best for: Labs with 50+ scientists who need to keep high-value instruments available, compliant, and efficiently used.
💡 Pro Tip: You don’t have to replace your current LIMS or ELN to use newLab. It’s designed to complement
2. Benchling
Best for Cutting-Edge Biotech R&D
Benchling has quickly become the gold standard for biotech R&D teams, especially those working with DNA, RNA, and protein design.
It combines Electronic Lab Notebook (ELN) and Laboratory Information Management System (LIMS) features in a sleek, collaborative cloud platform.
Key features
- Unified ELN + LIMS for experiment tracking & sample management
- DNA, RNA, and protein design tools
- Integrated inventory & workflows
- APIs and integrations with instruments & data analysis tools
Why labs choose Benchling
- Loved by scientists – intuitive, fast, and visually clear
- Designed for collaborative biotech R&D, ideal for teams scaling from 10 to 500+ researchers
- Strong compliance (21 CFR Part 11 electronic signatures, audit trails)
Best for: Fast-growing biotech startups or R&D divisions that want modern UX and biology-focused features.
3. Labguru
Best All-in-One Platform for Small to Mid-Size Labs
Labguru is a web-based ELN + LIMS + inventory system that’s known for its ease of use and fast onboarding. It’s especially popular with academic groups and smaller biotech startups.
Key features
- ELN with protocol templates
- Sample & reagent inventory tracking
- Equipment management (basic)
- Project & data organization
- Simple compliance: 21 CFR Part 11 support
Why labs choose Labguru
- Quick setup, intuitive UI
- Lower cost than enterprise systems
- Strong support and training resources
Best for: Labs with limited IT resources that want a budget-friendly, integrated solution.
4. LabWare LIMS
Best for Enterprise & Regulated Environments
For large-scale, compliance-driven biotech labs, LabWare is an industry heavyweight. It’s highly customizable and supports multi-site, multi-team operations.
Key features
- Comprehensive LIMS with ELN module
- Complex sample workflows & automation
- Full audit trails and compliance support (21 CFR Part 11, GLP, GMP)
- Integration with ERP and instrumentation
Why labs choose LabWare
- Battle-tested for enterprise and pharma
- Extremely configurable to match unique workflows
- Robust compliance validation
Best for: Big pharma and enterprise biotech companies with strong IT resources.
5. STARLIMS
Best for Deep Compliance & Analytics
Owned by Abbott, STARLIMS focuses on data integrity and compliance across highly regulated environments such as pharma, clinical, and diagnostics.
Key features
- Full LIMS with analytics & mobile apps
- Document & workflow management
- Comprehensive compliance (FDA, ISO, GMP)
- Integration with QC and manufacturing systems
Why labs choose STARLIMS
- Trusted in regulated pharmaceutical and diagnostics labs
- Strong mobile and analytics capabilities
- Global enterprise support
Best for: Labs where regulatory compliance drives purchasing decisions.
6. SciNote
Best Open-Source Option with Compliance Features
SciNote provides an open-source electronic lab notebook that’s FDA 21 CFR Part 11-ready, with optional paid hosting/support.
Key features
- ELN with structured projects and tasks
- Inventory & reagent management
- Electronic signatures and audit trails
- Integrations via API
Why labs choose SciNote
- Cost-effective, especially for startups & academia
- Open-source flexibility
- Used by FDA, USDA, and European Commission labs
Best for: Teams that need compliance-ready ELN features on a budget or want open-source control.
7. LabVantage
Best Enterprise LIMS + Analytics Platform
LabVantage is a premium enterprise informatics suite that combines LIMS, ELN, SDMS (Scientific Data Management), and analytics in one platform.
Key features
- Fully integrated informatics stack (LIMS + ELN + SDMS)
- Advanced analytics & dashboards
- Strong regulatory compliance & validation tools
- Modular deployment
Why labs choose LabVantage
- “Single source of truth” for all scientific data
- Excellent for multi-site, global organizations
- Tailored deployment and integrations
Best for: Large biotech/pharma companies looking for a comprehensive informatics hub.
How to Choose the Right Lab Management Software
- Map Your Workflows
- Where does data live?
- How are samples and instruments tracked today?
- Identify Compliance Needs
- FDA 21 CFR Part 11? ISO? GxP?
- Audit trail requirements?
- Evaluate Scalability
- Will the system handle 100+ scientists? Multiple sites?
- Will the system handle 100+ scientists? Multiple sites?
- Plan for Integrations
- ERP (SAP, Oracle), ServiceNow, analytics, instruments
- ERP (SAP, Oracle), ServiceNow, analytics, instruments
- Think Beyond Data: Equipment & Operations
- ELNs & LIMS rarely solve instrument scheduling or maintenance.
- Platforms like newLab can fill this gap.
- Test Before You Commit
- Request demos & pilot runs.
- Check user adoption – scientists should love it.
Why Adding newLab Is a Smart Move (Even If You Already Have a LIMS/ELN)
- Boost productivity: Automate instrument booking & maintenance.
- Reduce downtime: Proactive alerts & usage tracking keep equipment running.
- Improve compliance: Audit-ready logs for instruments and usage.
- Integrate with IT: Native to ServiceNow, so it fits into enterprise workflows.
- Scale with growth: Perfect for labs expanding R&D headcount.
Tip: Many labs run Benchling or LabWare for data but still rely on manual spreadsheets for equipment scheduling. That’s expensive and inefficient. Adding newLab eliminates this pain point while boosting ROI on your existing platforms.
Ready to modernize your lab operations?
Discover how newLab can integrate with your current ELN or LIMS to streamline equipment scheduling, compliance, and overall R&D efficiency.
👉 Book a 30-min demo with our team – see how to turn your lab into a seamless, AI-ready research environment.
Frequently Asked Questions
1. What’s the difference between an ELN and a LIMS?
An Electronic Lab Notebook (ELN) focuses on documenting experiments and research data, while a Laboratory Information Management System (LIMS) manages samples, workflows, and compliance tracking. Many modern platforms combine both.
2. Do I need both an ELN and a LIMS?
Not always, but many biotech labs use both. ELNs are better for scientific documentation, and LIMS handle sample logistics, QC, and compliance. Some solutions, like Benchling or Labguru, blend the two.
3. Is cloud-based lab software safe for regulated labs?
Yes – most modern platforms (Benchling, Labguru, newLab) offer secure, compliant cloud hosting with audit trails, access controls, and FDA 21 CFR Part 11 support.
4. How long does it take to implement a LIMS?
Small systems (Labguru, SciNote) can be running in weeks. Enterprise solutions (LabWare, STARLIMS, LabVantage) may take several months due to customization and validation.
5. Can newLab replace my LIMS or ELN?
No. newLab is designed to complement your current systems by solving equipment scheduling, maintenance, and operational gaps – not to replace data or sample management tools.
6. What if our lab already uses spreadsheets for scheduling? That’s common – but error-prone and hard to scale. newLab automates scheduling, adds compliance logs, and integrates with IT systems like ServiceNow.




